Locations/Addresses/Contact Info
Arterex operates through 16 state-of-the-art manufacturing facilities with 24 Clean Rooms and over 830K sq ft manufacturing space on 4 continents with 2,100 employees and growing. We are a global provider of diversified manufacturing solutions to the medical device, and life-sciences industries.
Arterex - Injection Molding, Tooling, Metals + Device Assembly
(Formerly Adroit USA)



Specialties: Complete Turnkey Solutions and Medical Device Fabrication, All Under One Roof
Locations:
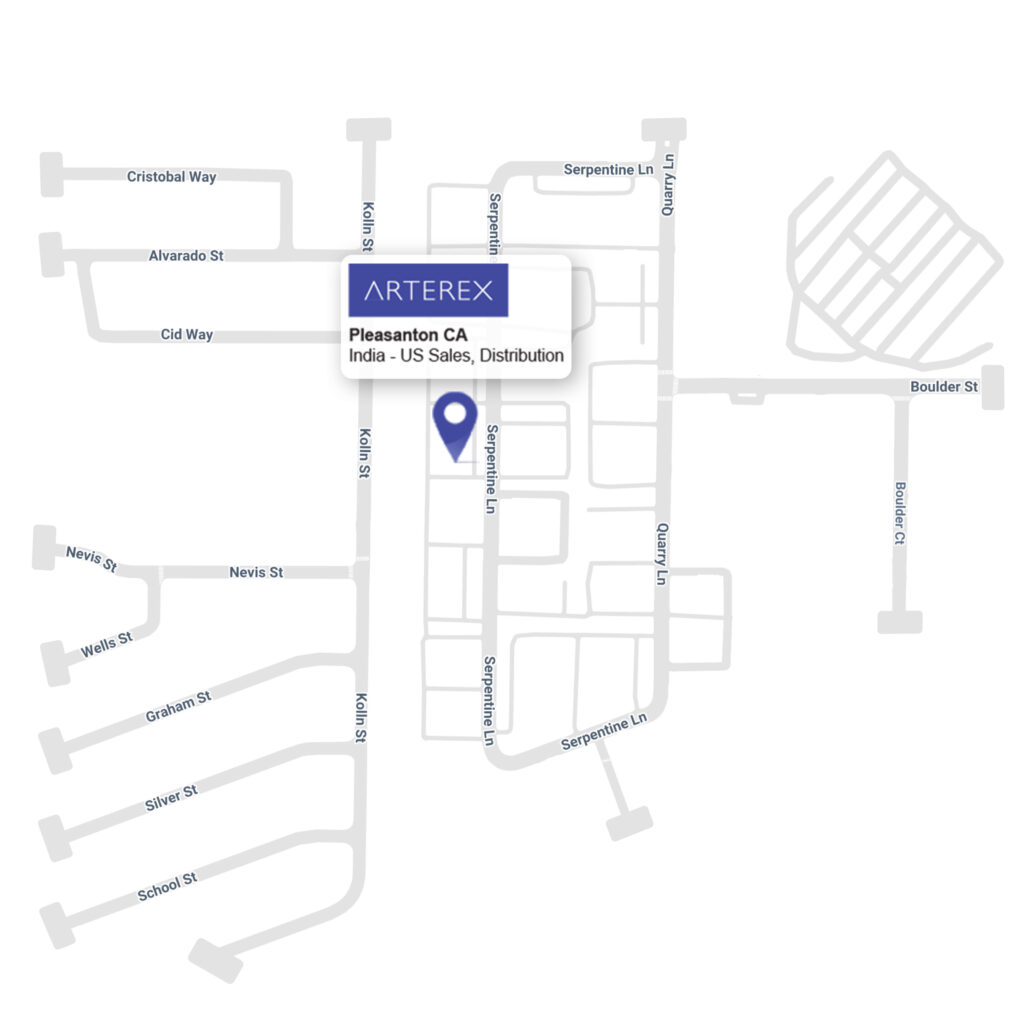
- Headquarters- 1043 Serpentine Lane, Suite A, Pleasanton, CA 94566
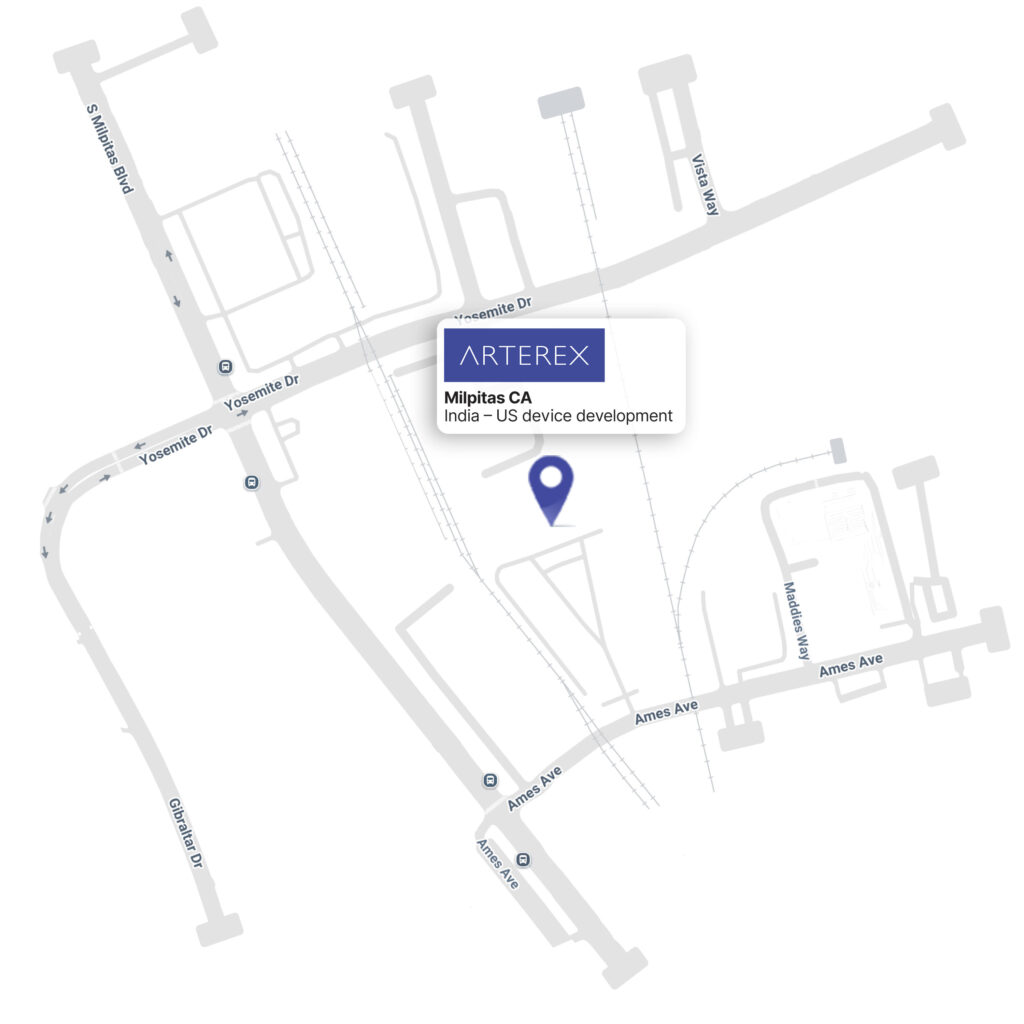
- Applications Lab — 773 Ames Ave, Milpitas, CA 95035
- Manufacturing Facilities in India
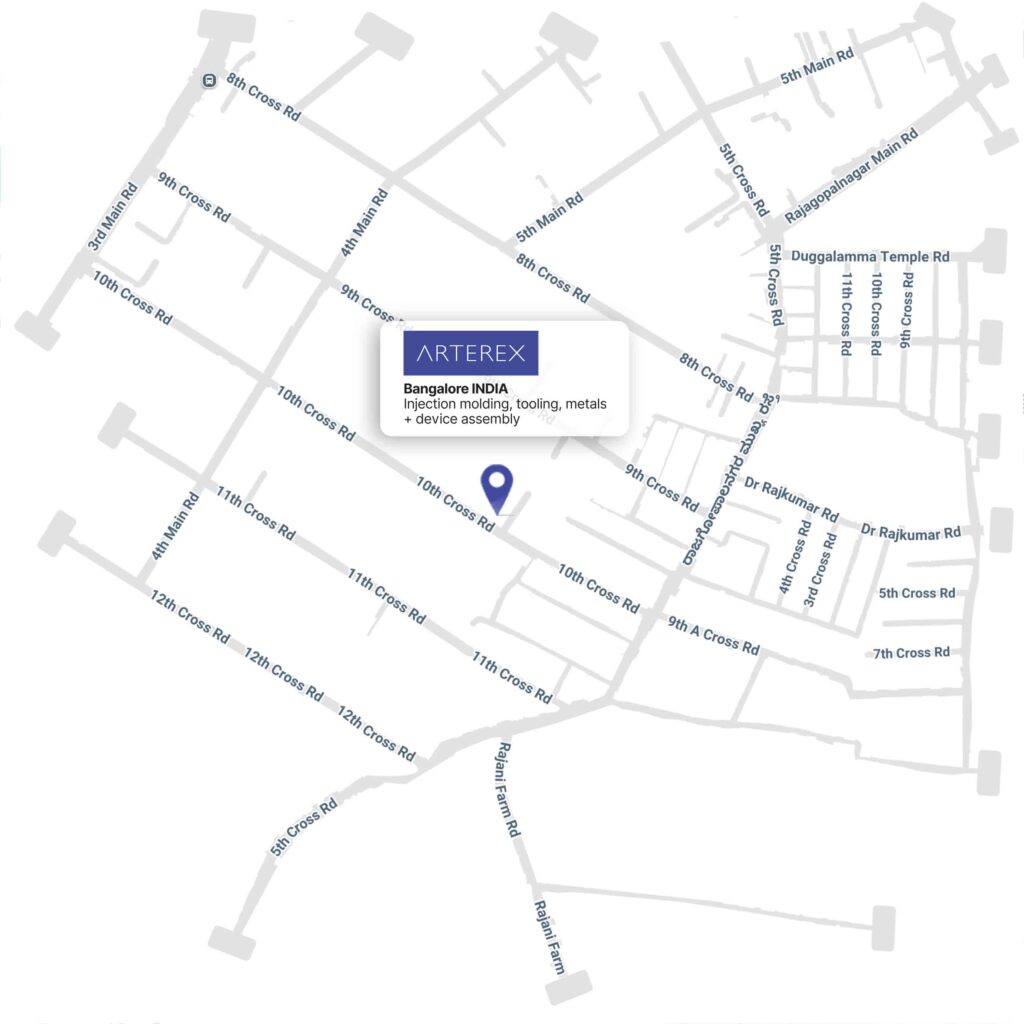
- Unit 1 — Plot No.375/A, 10th Cross, 4th Phase, Peenya Industrial Estate, Bengaluru, Karnataka – 560058, India
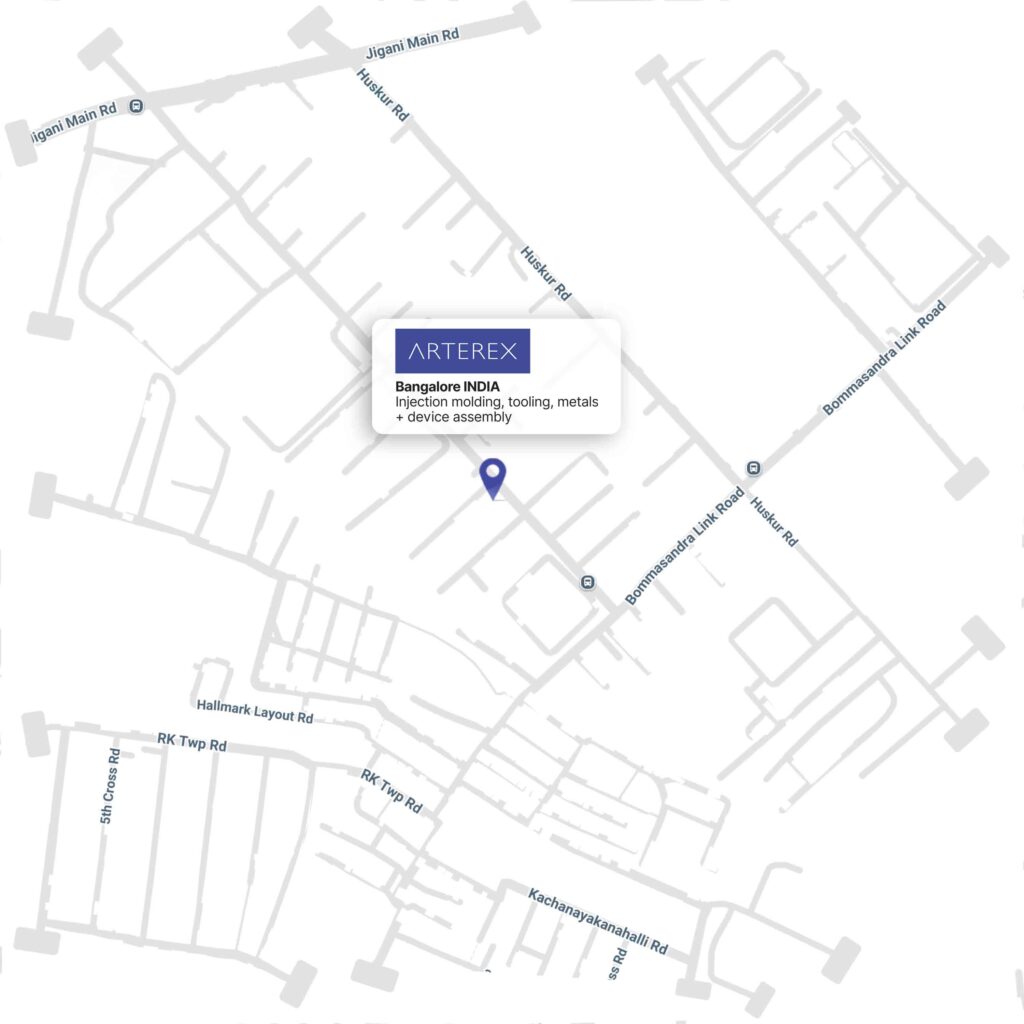
- Unit 2 — #15B, Bommasandra Industrial area, Hosur Road, Anekal Taluk, Bengaluru, Karnataka 560099, India
Standards & Quality Certifications: FDA Registered ISO 13485 certified, ISO Class 8 clean rooms for molding, ISO Class 7 clean rooms for assembly, FDA registered
From these locations in California and India, we provide product design & development and custom, value added, turnkey, component and finished device manufacturing solutions to the MedTech Industry from three facilities in Bangalore, India. Integrated manufacturing capabilities include extrusion, injection molding & tooling, comprehensive metals processing and finished device assembly, packaging and sterilization management.
Arterex - Injection Molding, Tooling, Device Assembly
(Formerly Formula Plastics)
Specialties: Medical Device Manufacturing, Plastic Injection Molding
Location: MX: Calle Guaycura No.106 Col, El Paraiso Tecate, BC 21485
This Arterex facility focuses on fully integrating molded plastic parts and other manufactured or purchased components into a finished and ready-for-use (sterile or non-sterile) medical device. With our ISO 13485:2016 quality systems and our process validation, we can ensure consistent product quality and delivery. Our capabilities include ISO Class 8 assembly and injection molding capacity, plastic joining and decorating technologies, including radio frequency and ultrasonic welding, solvent and UV bonding, and various decorating technologies. Part design and development for manufacturability – from prototype to mass production. This includes a full range of secondary operations such as assembly, welding, labeling, packaging, and more.
Arterex - Electromechanical Device Development + Device Assembly
(Formerly NextPhase)



Specialties: Complex Single-Use Devices (SUD), Electromechanical Devices (EMD), and Active Implantables for OEMS
Locations:
- Mansfield110 Forbes Boulevard
Mansfield, MA 02048
- Rochester88 Airport DriveRochester, NH 03867
- TijuanaBlvd. Tomas Alva Edison 14181,
Col. Tomas Alva Edison,
Tijuana, Baja California, Mexico
C.P. 22163
Standards & Quality Certifications: FDA Registered ISO 13485: 2016
From these three locations located in New England and Mexico, we have well-trained customer-centric teams with extensive medical industry knowledge and the drive to deliver the highest quality medical device design, development, manufacturing, and assembly services available. Arterex is a uniquely differentiated company with a proven track record for design, engineering, and manufacturing of complex single-use devices (SUD), electromechanical devices (EMD), and active implantables for OEM’s requiring a full-service contract manufacturing partner. Our more than twenty years of experience have earned us numerous industry awards, including Medical Design Excellence awards. We’re proud to have a range of customers spanning from innovative start-ups to multinational OEMs.
Arterex - Specialty Extrusion
(Formerly Xponent Global)
Specialties: High quality precise filaments, over the wire extrusion, multi-lumen, taper tubing, para tubing and tight tolerance single lumen tubing for a wide range of applications from simple packaging tubes to demanding products used in neurological and laparoscopic devices.
Location: 30 Tower St, Hudson, MA 01749, United States
Standards & Quality Certifications: ISO 9001:2015 certified manufacturer
Arterex offers a wide range of cleanroom extrusion capabilities from precise tight tolerance single lumen tubing to multi lumen and profiles. We offer paratubing and parawire extrusions and over extrusions of wires and metal profiles. We also have the capability for inline and offline secondary operations including punching, skiving, grinding, tipping, printing and overmolding.
Kabo, An Arterex Company

Specialties: Medical Bags and Medical Device Manufacturing & Research and Development
Locations:
- Kabo S.r.l
Via Partigiani d’Italia, 3643029 Traversetolo (PR) Italy - Kabomed L.L.Co.
Ind. District B4 – Area 174
10th of Ramadan City Egypt
Standards & Quality Certifications: ISO-9001-2018, ISO-13485-2012, Directive 93/42/ ECC
Kabo operates in the medical device sector and specializes in self-produced bags made of different materials and sizes for various needs. Our devices result from significant investments and solid partnerships with suppliers, which is part of our success. Each new project begins with a detailed feasibility study and an accurate assessment of its profitability. Our commitment to partnering with suppliers and continued cost analysis allows us to provide valuable products for our customers.
Luc & Bel, An Arterex Company
Specialties: Custom Components for Infusion, Hemodialysis, Cardiac Surgery and Transfusion, Research and Development
Location: Via Carlo Marx, 162, 41012 Carpi MO, Italy
Standards & Quality Certifications: ISO 80369-7, ISO 13485:2016, ISO Class 8 (EN ISO 14644-1)
LUC & BEL designs and produces excellent quality and reliable components for medical devices. Its teams’ experience, combined with the latest production technology, offers a wide range of first class engineering solutions. Our products perfectly fulfill all our customers’ requirements, and we are among the world’s top medical suppliers of our products. Luc & Bel uses state-of-the-art production technologies to offer high-quality medical products. These range from individual components to complete devices based on the needs of each customer and to specific sectors such as infusion, hemodialysis, cardiac surgery and transfusion.
ModenPlast, An Arterex Company

Specialties: Medical Bags, Research and Development
Location: Via Montegrappa, 24/26
41042 Ubersetto di Fiorano
Standards & Quality Certifications: ISO Class 8 (EN ISO 14644-1), ISO 13485, ISO 9001
ModenPlast offers medical-grade PVC production for extrusion and injection molding. ModenPlast specializes in the extrusion of PVC and PVC DEHP FREE tubing, produced with its compound named “RB” and EVA, TPU, TPE, and PE. Our company can also extrude lay-flat tubing in PVC and EVA to produce medical bags. This spectrum gives both Modenplast and all its customers, the possibility to work in a large application field.
Phoenix, An Arterex Company
Specialties: Design, Development, and Manufacture of Disposable and Non-Disposable Medical Devices
Location: Via Leonardo da Vinci, 55 San Felice S/P, 41038 Modena, Italy
Standards & Quality Certifications: ISO 13485:2016
Phoenix is a leading European medical device developer and manufacturer and provides a wide range of services related to the design, development, and manufacture of disposable and non-disposable medical devices. Offering product development, production, assembly and packaging services.
Synecco, An Arterex Company

Specialties: Medical equipment Design & Contract Manufacturing Solutions
Locations:
- Design Centre
Block 12, Galway Technology Park
Parkmore, Co. Galway, H91 E4YD - Manufacturing Site
Scariff Road, Whitegate
Co. Clare, V94 FA03
Standards & Quality Certifications: NSAI (EN ISO 14001:2015), NSAI (EN ISO 13485:2016)