Arterex Unveils Unified Brand Identity Across North America, Delivering a More Consistent Global Customer Experience
Written by
Arterex Medical
Published on
January 20, 2026
Strategic regional brand architecture enhances clarity, scale, and customer alignment
(Arterex, January 20, 2026) — Arterex, a leading global contract medical device design, development, and manufacturing partner, today announced the evolution of its brand identity, reflecting the company’s continued growth and commitment to delivering integrated, high-quality solutions to customers worldwide.
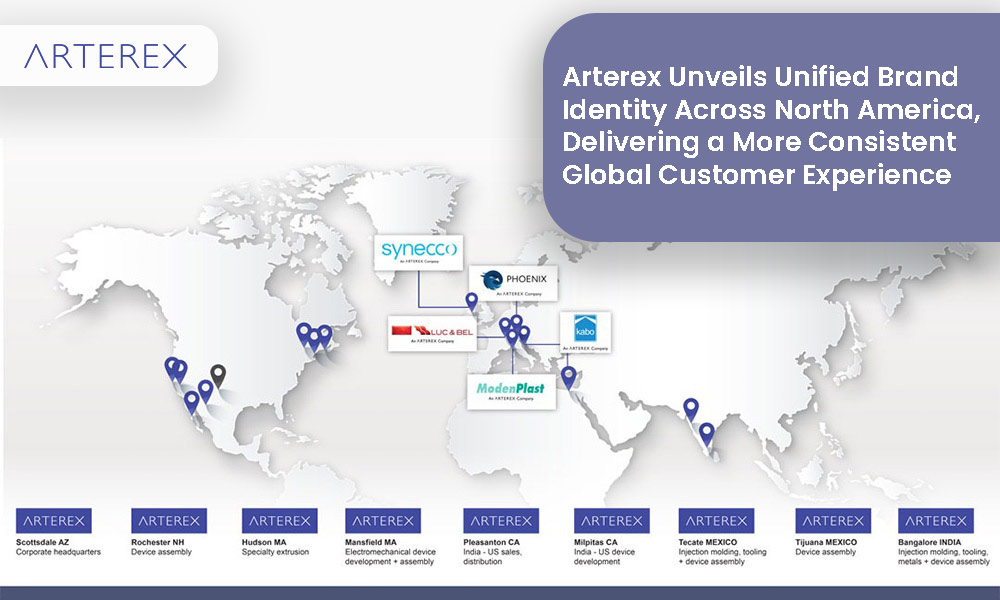
As part of this evolution, Arterex’s North America–based operating divisions will transition to operate under the Arterex name and visual identity, creating a unified presence across the region. This move simplifies engagement, strengthens brand recognition, and reinforces Arterex’s position as a cohesive, end-to-end partner for medical device innovators.
In Europe, Arterex’s Italy- and Ireland-based operating companies will retain their established local brand identities, reflecting their deep regional heritage, longstanding customer relationships, and strong regulatory reputations. These businesses will continue to operate as integral members of the Arterex group, fully aligned with Arterex’s global quality systems, operational standards, and strategic direction.
“This brand evolution is intentionally regional and customer-driven,” said Jeff Goble, CEO of Arterex. “In North America, a unified Arterex brand brings clarity, scale, and speed. In Europe, our operating companies have earned exceptional trust under their existing names, and maintaining that equity best serves our customers.”
By consolidating branding across North America, Arterex enhances consistency, reduces complexity, and enables customers to more easily access the full breadth of the company’s capabilities. At the same time, Arterex’s European businesses remain empowered to leverage their strong local identities—now clearly endorsed and supported by the global Arterex platform.
“This transformation is more than a visual change,” Goble added. “Whether operating under the Arterex name or a long-established local brand, every customer receives the same commitment to quality, innovation, and collaboration—backed by the full strength of Arterex.”
Arterex operates 16 state-of-the-art facilities across four continents and employs more than 2,100 professionals worldwide. The company provides comprehensive design, development, engineering, compounding, extrusion, injection molding, blow molding, metals processing, tooling and mold building, and advanced assembly services to leading medical device and life sciences companies.
The updated Arterex brand identity will roll out across North America beginning January 20, 2026, with corresponding updates to digital platforms and customer communications.
For more information, visit www.arterexmedical.com or contact inquiries@arterexmedical.com.

For further information please contact:
S!Y Communications, Inc.
Andrea Siy, President
Tel: +1 (978) 270-4080
Email: Andrea@siycommunications.com
About Arterex
Arterex is a multi-capability medical device developer & contract manufacturer resulting from the combination of Kabo, Formula Plastics, Luc & Bel, ModenPlast, NextPhase, Micromold, Phoenix, Adroit USA, Xponent Global and Synecco. The company is now branded in North America as Arterex and serves a wide range of customers, from blue chip global MedTech OEMs to leading innovators. The Arterex companies headquartered in Italy (Kabo, Luc & Bel, Modenplast and Phoneix), and in Ireland (Synecco) have all maintained their existing brands. Arterex has a global manufacturing footprint with sixteen state-of-the-art facilities across the US, Mexico, Italy, Ireland, Egypt, and India.
Additional information is available at www.arterexmedical.com.
